The increasing awareness of society about nature conservation and the recovery of natural spaces has put pressure on governmental bodies to tighten up the laws on pollution prevention and control; for instance, Directive 2008/1 of 15 January on Integrated Pollution Prevention and Control, and private corporations in order to minimise the impact of their activity.
The iron and steel industry in general, and the metal surface finishing industry in particular, are facing great challenges to reduce the impact of their production processes. These production processes include the pickling process. This process is used to eliminate the oxide layer that covers metal parts in order to perform subsequent processes (painting, metal coating, etc.)
In the 1960s, the use of hydrochloric acid solutions as a pickling bath for steel surfaces was widespread due to its low cost and efficiency at room temperature. The oxide and other impurities that are eliminated during the pickling process are dissolved in the bath in the form of iron chloride (FeCl2) and affect the efficacy of the pickling.
The pickling bath is considered spent when the iron chloride solubility limit is reached (∼ 150 g/L). The spent pickling baths lack the capacity to remove oxide and impurities from the metal surfaces and must be renewed.
The spent baths are the greatest environmental problem facing metal surface finishing industries and they must be disposed of by authorised waste managers at a high cost.
The presence of zinc generated by the galvanising industry in the spent pickling baths is an additional problem in the management of the effluent, as not all recovery processes are able to work with zinc chloride (ZnCl2).
It is common practice for the waste managers entrusted with eliminating the effluents from the surface finishing and galvanising industries to neutralise the spent pickling baths with alkaline substances to precipitate metal hydroxides. This is an expensive process that presents landfill issues due to the high salinity of the treated effluent, generating huge quantities of sludge –which must be neutralised– and there is no recovery of raw materials or recoverable by-products.
The commitment to protect the environment, provide inexpensive, sustainable solutions and the recovery of raw materials forms part of the mission of Condorchem Envitech, S.L. For this reason, the organisation has embarked on the CHEMIREC® project, a system for the recovery of minerals and recoverable elements from spent pickling baths in order to develop an extremely innovative solution for the recovery of hydrochloric acid from the effluent of spent metal pickling baths.
Unlike current techniques for the treatment of spent pickling baths, the CHEMIREC® system permits the complete recovery of both free and combined hydrochloric acid to obtain ferrous sulphate (FeSO4) as a commercially recoverable by-product for use as a fertiliser and in the case of the galvanising industry, the recovery of zinc chloride for use in medical practice.
So, the execution of the CHEMIREC® project constitutes a highly effective solution to an environmental problem that has not yet been solved and is derived from the widespread global practice of treating metal surfaces to protect them from corrosion. The process permits the recovery of more than 95 % of the hydrochloric acid present in spent pickling baths, with a low free acid content (1 – 10 %), cost savings in raw materials thanks to the reuse of the recovered hydrochloric acid and zinc chloride (in the case of the galvanising industry), and the commercial recovery of ferrous sulphate in the form of a fertiliser.
The purpose of this phase is to eliminate all the substances that make it impossible to reach the ultimate objective. The pickling bath contains suspended particles from the impurities introduced into the process when the parts are placed in the pickling bath. In addition, the presence of oils and surfactants has been detected. The oils come from the parts themselves, whereas the surfactants are an additive that is incorporated into the pickling solution to improve its wetting power and accelerate the pickling process. These particles, oils and surfactants are harmful to the system, and so a filtering system has been designed to eliminate these particles.
Once these elements have been eliminated from the bath, it passes to a liquid-liquid extraction system in which it is put into contact with an organic extractant. The mission of this extraction process is to eliminate the zinc contents from the initial bath, transporting the extractant in the form of zinc chloride.
This step makes it possible to recover the zinc present in the spent pickling bath. Zinc is a very valuable metal that can be reused in the production process.
To recover the zinc and regenerate the extractant, a new liquid-liquid extraction of the zinc present in the organic extractant to ultrapure water is performed. This allows us to obtain a pure extractant that can be reused and a zinc chloride dissolution that can be recovered or reused in the process as a fluxing agent.


System for the recovery of zinc from the spent pickling bath installed in a shipping container.
The purpose of this phase is to eliminate as much iron as possible from the bath. To that end, sulphuric acid is added to it and the mixture is cooled, which provokes the crystallisation of the ferrous sulphate. To carry out this process, the bath (without zinc) and the sulphuric acid are inserted into the crystalliser, which consists of a tank with a cooling coil and a gentle stirring system to homogenise the temperature. A chiller is used to cool the bath.
Once the ferrous sulphate salts have formed, they are filtered and dried in a basket centrifuge built of special acid-resistant material.
Once the zinc and iron content of the bath has been reduced and it has a large quantity of chlorides and free acidity, as is the case after crystallisation, it is distilled using the membrane distillation technique. This distillation is arranged in such a way that the concentrate is recirculated until the quantity and characteristics of the distillate match those calculated for the mother liquor concentrations.


System for the crystallisation and recovery of hydrochloric acid
This equipment was installed in two shipping containers in order to facilitate its assembly, location and transfer.

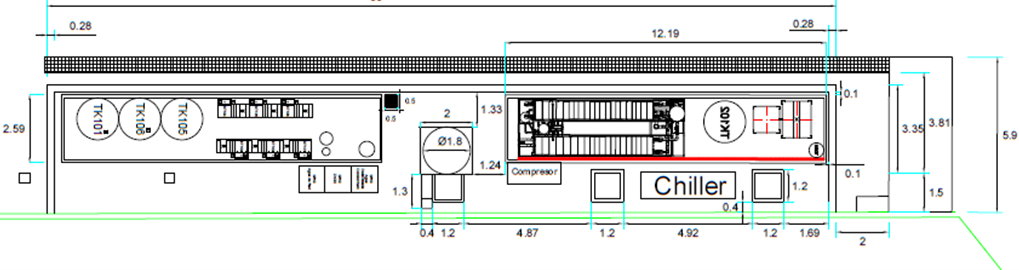
General view
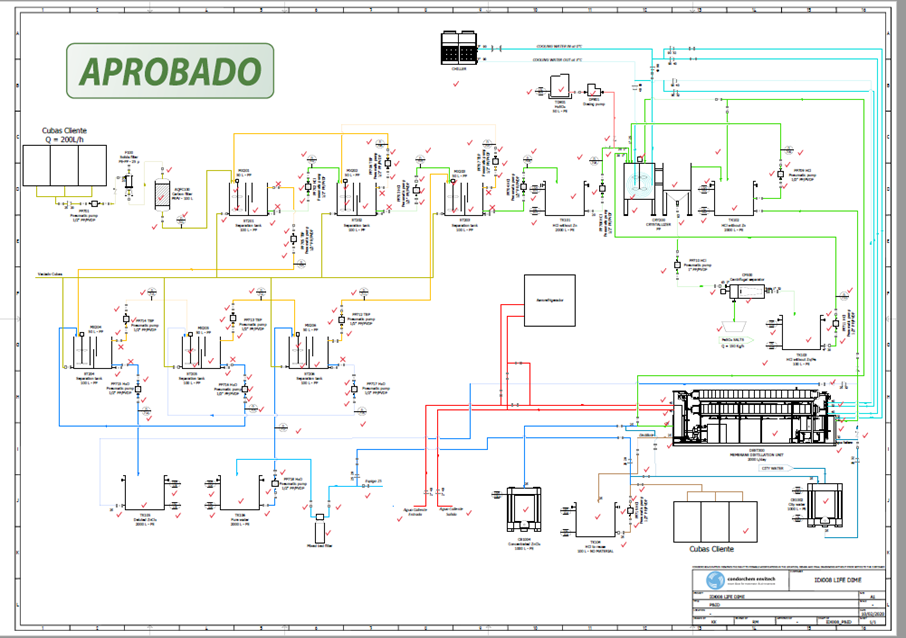
Operating diagram
Once the mechanical and electrical assembly of the plant has been completed, it was put into operation. During the start-up, a series of operating tests were performed in accordance with a production sequence and progressive adjustments to the equipment and system. The objective of the pilot plant start-up was to ensure its operation in accordance with production output requirements, while respecting the legally-established environmental, security and hygiene specifications.
The tests performed are described below:
As described above, the spent bath contains suspended solids, surfactants and separated oils. These products are harmful to the process and so the first step taken was to filter the spent pickling bath.
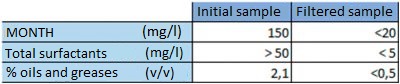
Table showing the values obtained after filtering
The presence of zinc in the acid recovery process is not a problem and there is no need to eliminate it in the pickling bath recovery process. However, the zinc will precipitate in the form of zinc sulphate together with ferrous sulphate, contaminating the latter. Furthermore, the fact that the zinc is not recovered would mean an important loss of raw materials.
Therefore the zinc is recovered with the aim of being able to recover the ferrous sulphate obtained during the process and recover a raw material that is used in the galvanising process.
The zinc is eliminated by means of a liquid-liquid extraction, using an organic phase. The extractant is an organic solvent that is often used in the metal extraction and purification industry.
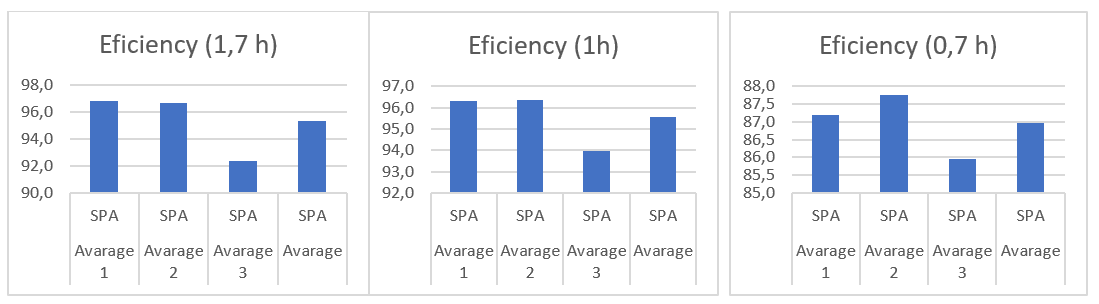
Efficiency of the Zn extraction system at different contact temperatures
Zinc is a raw material that can be reused in the galvanising process if it is present in the adequate concentration and in the aqueous phase.
In addition, the extractant product has a high cost, and so it is useful to regenerate it in the process. Following laboratory tests, it was observed that the best system to recover it is by performing a cascade extraction with ultrapure water.
The ultrapure water is produced in the CONDORCHEM ENVITECH facilities by mixed bed ion exchange resins equipment.
The system used is the one described above, but using ultrapure water instead of a bath.
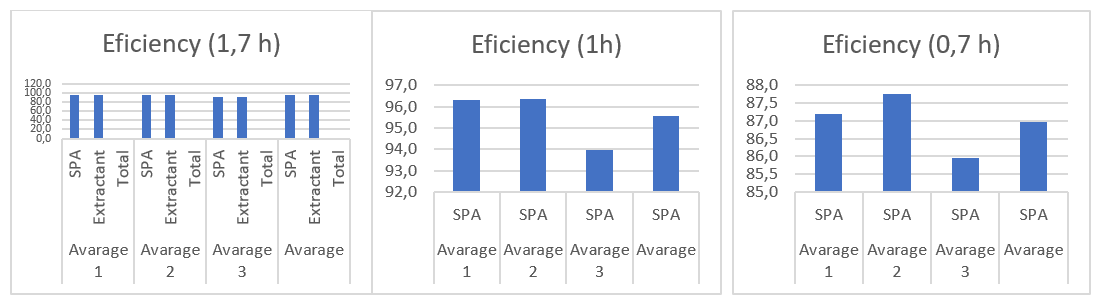
Efficiency of the Zn extraction system at different contact temperatures
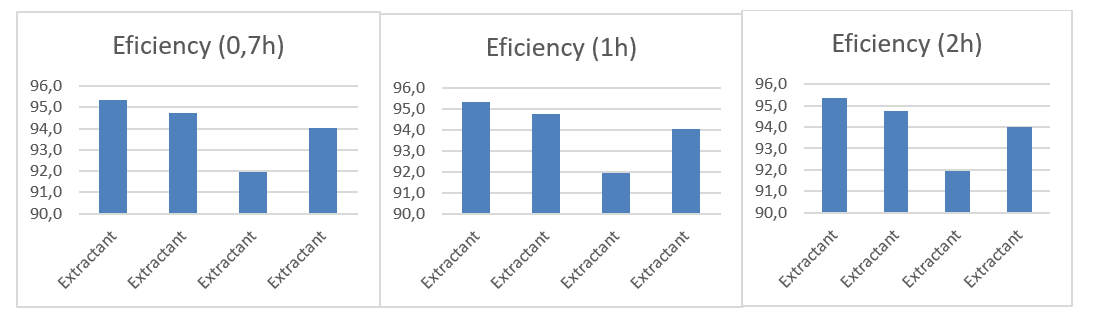
Efficiency of the Zn extraction system at different contact temperatures
Work is currently in progress on this aspect on a pilot plant scale, but the pilot plant scale results obtained are described below.
The evaporator concentrate from the previous system is added to the pre-treated bath for the purpose of crystallising the concentrated ferrous sulphate and recovering the maximum possible concentration of hydrochloric acid. Sufficient sulphuric acid is added to the sample to precipitate the divalent iron and obtain the maximum free acidity to recover the combined hydrochloric acid.
Once this mixture has been obtained, it is placed inside the crystallisation reactor and the temperature is lowered to 2 – 4 ºC. After reaching this temperature, it is left to decant for 30 minutes and the reactor is emptied into a filter bag that separates from the bath the ferrous sulphate crystals with a low iron content.
The results obtained in preparing the different extractions were as follows:
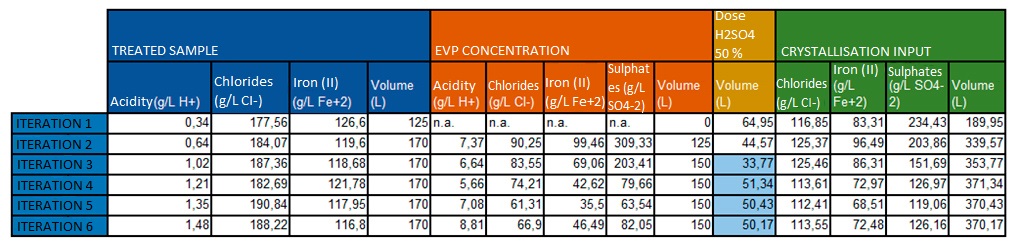
In the first two samples, an excess quantity of sulphuric acid was inserted in order to increase the quantity of sulphates present in the medium and shift the balance to the precipitation of ferrous sulphate salts. In the following iterations, it was performed stoichiometrically.
The results of the extraction are shown in the following table:
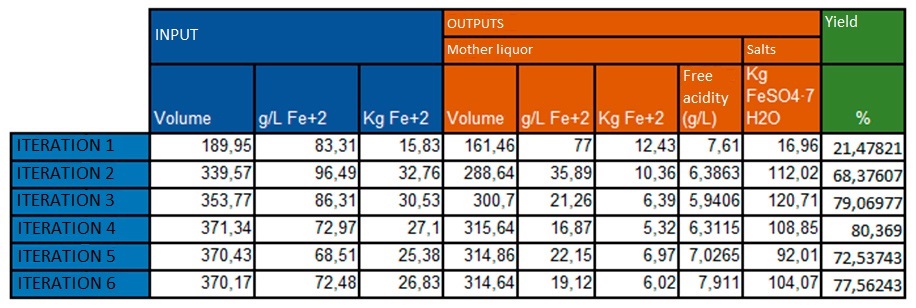
The outputs are adjusted to the expected values, observing a tendency for the divalent iron output to remain constant in the mother liquor.
The mother liquor extracted from the crystallisation system is placed in the membrane evaporation equipment (described earlier). It is widely documented in literature that on distilling a solution that contains hydrochloric acid, an azeotrope is formed that will have a hydrochloric acid concentration of almost 18 %:
For this reason, it is not likely that hydrochloric acid concentrations above this value will be obtained.
The content of chlorides in the mother liquor at the crystalliser outlet is around 120 g/L. In addition, based on previous laboratory tests, it was seen that the hydrochloric acid concentration in this distillate is around 150 g/L of hydrochloric acid.
This entails the need to add a volume of water that guarantees the recovery of the maximum quantity of hydrochloric acid. It has been determined that each iteration will be supplemented with water up to 330 litres, except for the first, as no concentrate is added from previous cycles and it will be supplemented up to 225 litres.
The results obtained are set out in the following table:
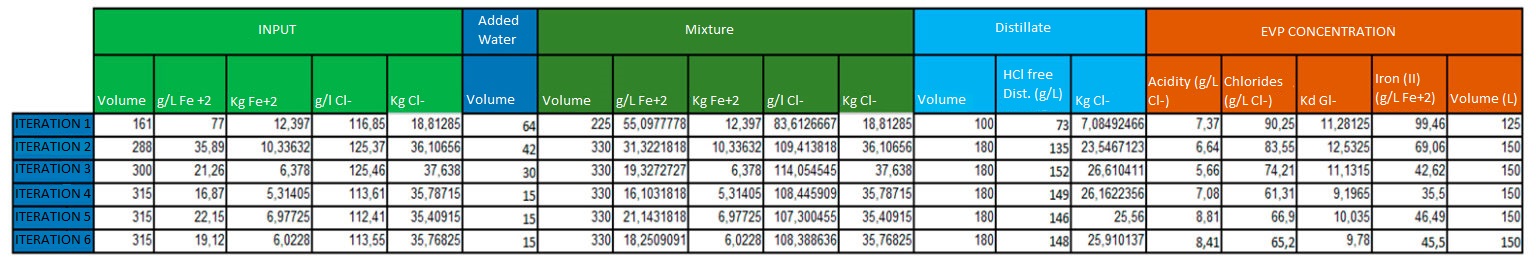
It is observed that the values stabilise at a concentration of 150 g/l of hydrochloric acid in the distillate and a concentration of 65 g/l of chlorides in the concentrate.
The individual result of each step is explained below:
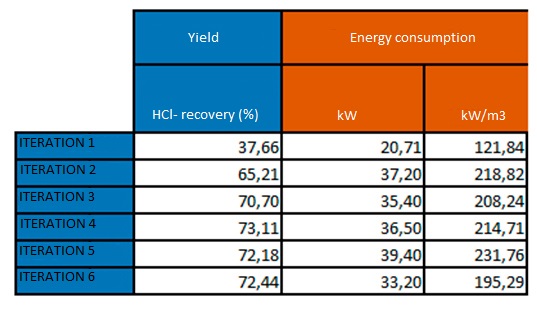
Zinc can be reused in the plant itself, or it can be sold as a by-product to companies that formulate chemicals for the galvanising industry.
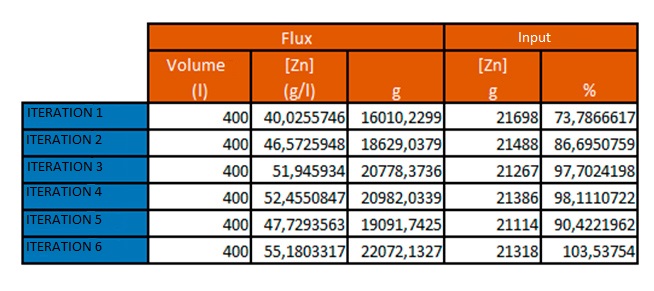
The zinc chloride present in the water is very diluted and can be reused or resold. For this reason, a preliminary study has been conducted on the possibility of reconcentration within normal parameters for its sale or reuse.
This test was performed on a single phase, as the concentration process using DESALT equipment is well known by the company.
The results obtained are:
A3 VALIDATION OF THE TECHNICAL SOLUTION
The objective of this activity was to optimise the process for the treatment of the effluent of waste from spent pickling baths through a technological solution based on an extremely innovative process through patent P200400694, owned by CONDORCHEM. The operational parameters that were validated in this activity are:
All these parameters were evaluated experimentally in the pilot plant.
LIFE DIME solution has been successfully tested and completed as a process to recover and reuse valuable materials from spent pickling liquor. The initial goals have been achieved, which means that the LIFE DIME solution has been fully transferred to real world applications within the metal surface treatment industry.

LIFEDIME is a solution that recovers metals and hydrochloric acid from spent pickling baths.

LIFE DIME is co-financed by the LIFE program of the European Union. (Grant Agreement No. LIFE16 ENV/ES/00041). This website expresses only the opinion of the author hence the agency is not responsible for any possible use of the information herein contained.
© 2021 – LIFEDIME – Legal notice – Privacy Policy – Social Media Policy
